- Research
JCAP Researchers Integrate Theory and Experiment to Discover Novel Photoanodes and Pave the Way for Materials-by-Design Techniques
JCAP’s high-throughput experimentation and theory teams develop a means for rapid identification of the most promising classes of photoelectrochemical materials.
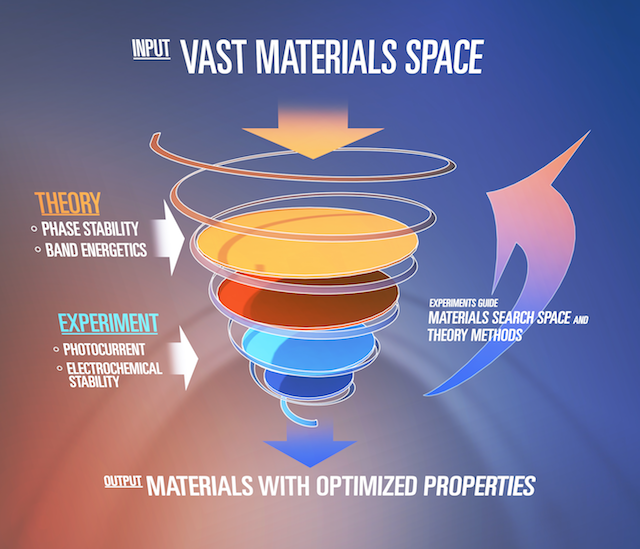
The promise of generating renewable energy from sunlight, carbon dioxide, and water can be realized through the discovery of efficient and robust photoelectrochemical (PEC) cell materials, specifically photoanodes. In an integrated solar-fuels device with tandem light absorber PEC architecture, the ideal photoanode candidates are semiconductors, which have band gaps between 1.6 and 2 eV and a valence band energy that allows efficient photoelectrocatalysis of the oxygen evolution reaction (OER). OER is an anodic reaction that is paired with two cathodic fuel-forming processes: hydrogen evolution and CO2reduction reactions. In collaboration with the Materials Project, JCAP’s high-throughput experimentation team, led by John Gregoire (Caltech), and a theory team, led by Jeff Neaton and Kristin Persson (LBNL), now have a defined means for rapid identification of the most promising classes of photoanodes. The results of this collaborative work have been reported in the latest issue of Advanced Energy Materials.
Employing a strategic combination of detailed electronic structure calculations, combinatorial materials synthesis, and both traditional and high-throughout photoelectrochemistry measurements, the JCAP team identified earth-abundant copper and manganese vanadate complex oxides that meet highly demanding requirements for photoanodes: low band gap energy, stability under highly oxidizing conditions, and valence band alignment with respect to OER. In the case of copper vanadates, the team pinpointed four distinct photoelectrocatalytic phases, which have the desirable band gap energy at or below 2 eV: α–Cu2V2O7, β–Cu2V2O7, γ–Cu3V2O8, and Cu11V6O26. In addition to showing excellent photocatalytic stability in electrolyte solutions with a pH as high as 13, these four phases appear to have important similarities in electronic structure, according to first-principles calculations.
“There are not many known photocatalysts capable of oxygen evolution, and this work expands the space of such materials in non-incremental fashion.”
—Jeff Neaton
JCAP’s integrated theory–experiment effort capitalized on the vast materials database and computational tools within The Materials Project. In the spirit of the Materials Genome Initiative, JCAP developed a workflow to evaluate materials using key parameters, such as band gap energy, band positions, and stability, under operational conditions. This computational effort carved out specific regions of the vast metal oxides materials space, which could be rapidly evaluated using high-throughput experimentation synthesis and screening techniques. Throughout the course of this study, the teams compiled an extensive library of experimental and theoretical data including electronic, magnetic, optical, photoelectrochemical, and structural properties, which are now used as feedstock in material genome work and near-term development of superior PEC materials through materials-by-design techniques. The comparison of theoretical and experimental results motivates researchers to conduct additional higher-accuracy calculations and measurements that yield a more complete understanding of the materials and how their structure and composition lead to remarkable photoelectrochemical performance.
JCAP researchers are very encouraged by the success of their theoretical work and experimental synergistic efforts and the positive feedback loop that is now established between the JCAP high-throughout experimental pipeline and parameter-free, first-principles theory. John Gregoire notes, “The importance of these results demonstrates that the allure of efficiency and speed offered by high-throughput methods can be realized while maintaining the required standard of experiment quality to produce top-notch science.” The team is also excited that the collective effort provides not only the discovery of high-performance materials, but also the advancement in scientific understanding of metal oxide photoanodes.
Jeff Neaton is enthusiastic about the contributions that this work imparts on the solar-fuels community and the broader materials science community. “There are not many known photocatalysts capable of oxygen evolution, and this work expands the space of such materials in non-incremental fashion,” Neaton says. “Additionally, the tight experiment-theory feedback loop has broader implications for materials theory. The close collaboration with experiment is not only teaching us about the efficacy of common approximations in electronic structure theory, but is also leading to important insights into the ways we might improve such approximations and consequently our ability to predict and design new materials.”
—Written by X. Amashukeli
This work is performed by the Joint Center for Artificial Photosynthesis, a DOE Energy Innovation Hub, supported through the Office of Science of the U.S. Department of Energy (Award No. DE-SC0004993). Computational work is supported by the Materials Project Predictive Modeling Center (EDCBEE) through the U.S. Department of Energy, Office of Basic Energy Sciences, Materials Sciences and Engineering Division, under Contract No. DE-AC02-05CH11231. Work at the Molecular Foundry was supported by the Office of Science, Office of Basic Energy Sciences, of the U.S. Department of Energy under Contract No. DE-AC02–05CH11231.
Publications:
- Yan, Q. et al. Mn2V2O7: An Earth Abundant Light Absorber for Solar Water Splitting. Advanced Energy Materials, DOI: 10.1002/aenm.201401840 (2015).
- Zhou, L. et al. High Throughput Discovery of Solar Fuels Photoanodes in the CuO–V2O5 System. Advanced Energy Materials, DOI: 10.1002/aenm.201500968 (2015).